APC, antigen-presenting cell BCR, B cell receptor circRNA, circular ribonucleic acid IRES, internal ribosome entry site IVT, in vitro translation LNP, lipid nanoparticle MHC, major histocompatibility complex mRNA, messenger ribonucleic acid saRNA, self-amplifying ribonucleic acid SARS-CoV-2, severe acute respiratory syndrome coronavirus 2 S protein, spike protein TCR, T cell receptor UTR, untranslated region. Immunization progresses with CD4 + helper T cells further helping in (1) activation of B cells that produce SARS-CoV-2 neutralizing antibodies and (2) activation of CD8 + cytotoxic T cells that may specifically recognize and eliminate virus-infected cells. 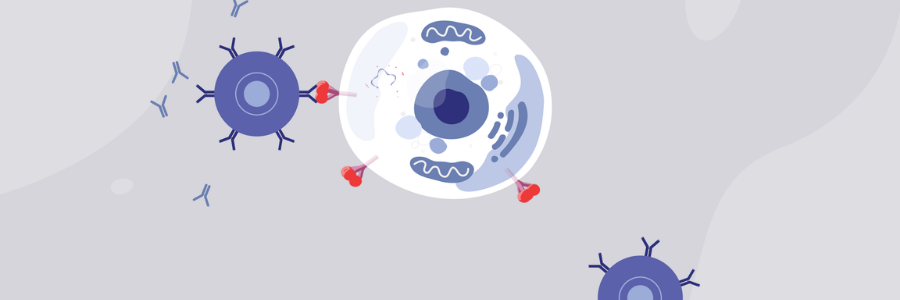 
Finally, the antigen protein can be exported from the cell and endocytosed back to the same or another APC, degraded by endosomal proteases, and presented on MHC II structures resulting in a CD4 + helper T cell response. Antigens can also be anchored to the membrane of the APC and directly recognized by BCRs leading to B cell responses however, such a path and its contribution to antibody production is currently under debate. The S protein is produced by the translational machinery of the APCs (red circles), degraded by proteasomes (pink circles), and presented on MHC class I (pink circles), leading to a specific CD8 + cytotoxic T cell response against SARS-CoV-2. ,, Antigen-encoding mRNAs are formulated into LNPs, endocytosed, and released through the process of endosomal escape to the cytoplasm. circRNA for vaccine application consists of a covalently closed single-stranded RNA that contains antigen sequence and an IRES that allows initiation of antigen translation. saRNA consists of the same sequence organization, but in addition contains: (1) a sequence encoding four non-structural proteins (nsP1–4), which form a replicase responsible for amplification of the saRNA, and (2) a subgenomic promoter (black arrow) of viral origin that initiates transcription of antigens. Depending on the use of native or modified nucleosides during IVT, unmodified or modified mRNAs are produced. The linear non-replicating mRNAs consist of a sequence encoding an antigen (e.g., the S protein for SARS-CoV-2) flanked by 5′ and 3′ UTRs, with a cap structure at the 5′ end and a poly(A) tail at the 3′ end. 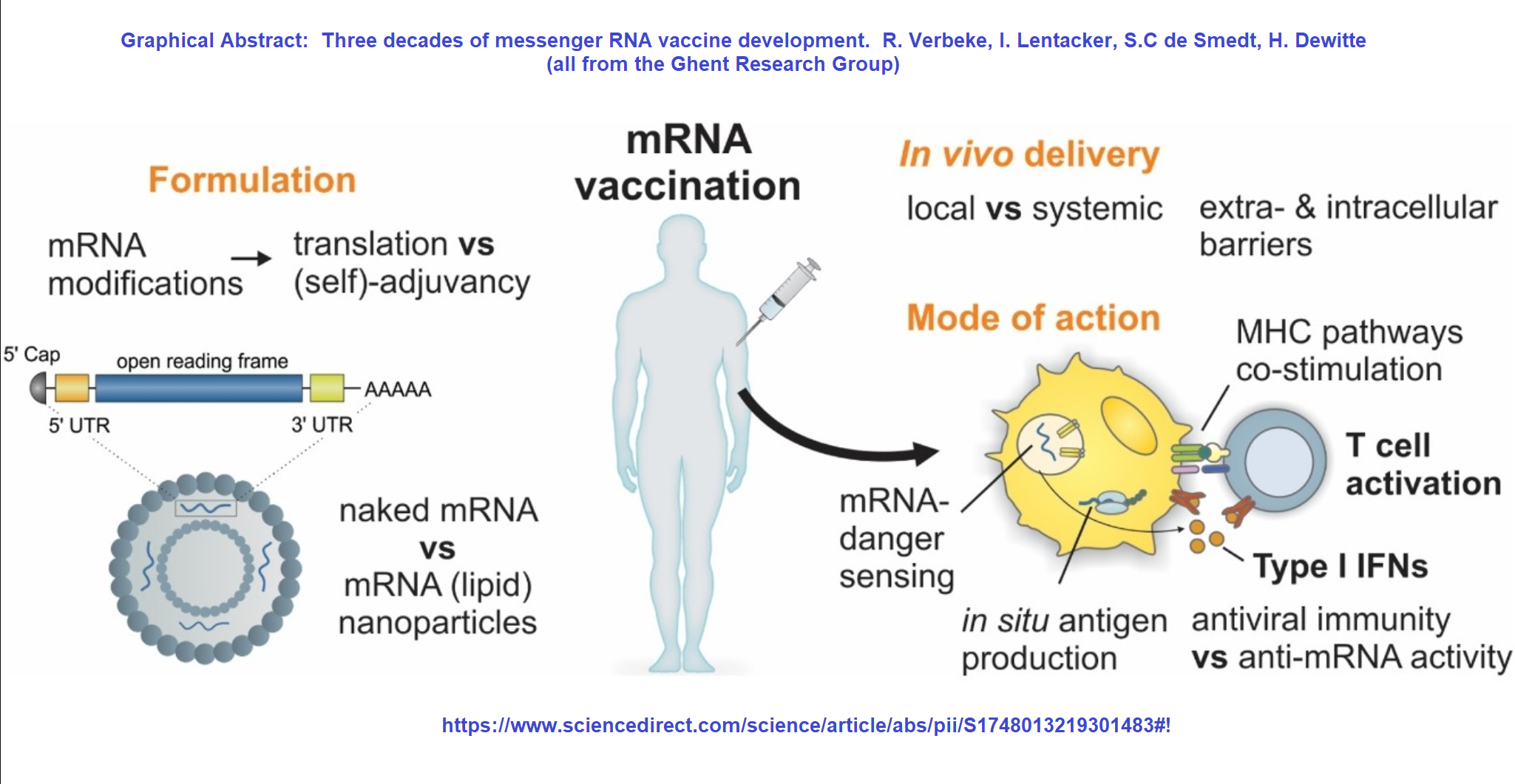
Immunization against COVID-19 with mRNA vaccines Immunization with mRNA vaccines requires an antigen-encoding mRNA transcript. We also discuss the current and anticipated challenges of these technologies, which may be important for future research endeavors and clinical applications.ĬOVID-19 first mRNA vaccine approval mRNA vaccine platforms mRNA vaccines nucleoside-modified mRNA.Ĭopyright © 2022 The Author(s). This review presents a brief history and the current status of development of four mRNA vaccine platforms, nucleoside-modified and unmodified mRNA, circular RNA, and self-amplifying RNA, as well as an overview of the recent progress and status of COVID-19 mRNA vaccines. This unprecedented success also emphasized the broader potential of this new drug class, not only for other infectious diseases, but also for other indications, such as cancer and inherited diseases. Real-world evidence following the deployment of global vaccination campaigns utilizing mRNA vaccines has bolstered clinical trial evidence and further illustrated that this technology can be used safely and effectively to combat COVID-19. Indeed, the first two vaccines against COVID-19 to receive accelerated regulatory authorization were nucleoside-modified mRNA vaccines, which showed more than 90% protective efficacy against symptomatic severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection alongside tolerable safety profiles in the pivotal phase III clinical trials. This research uncovered the unique favorable characteristics of mRNA vaccines, including their ability to give rise to non-toxic, potent immune responses and the potential to design and upscale them rapidly, making them excellent vaccine candidates during the coronavirus disease 2019 (COVID-19) pandemic. Since the first successful application of messenger ribonucleic acid (mRNA) as a vaccine agent in a preclinical study nearly 30 years ago, numerous advances have been made in the field of mRNA therapeutic technologies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
